
A Translational Science Benefits Model Case Study
Project in a Nutshell
Uterine fibroids are common reproductive-age tumors. More than 80% of Black women and nearly 70% of White women have fibroids by age 50. Currently available interventions are either expensive, difficult to access, or have significant systemic side effects.
Thanks to a collaborative partnership between Duke and North Carolina Central University (NCCU) researchers developed and tested a protective carrier for drug therapeutics to enable a direct local injection into the uterine fibroid. This presents
a minimally invasive treatment option for women suffering from uterine fibroids.
Significance of the Project
Uterine fibroids are common reproductive-age tumors. More than 80% of Black women and nearly 70% of White women have fibroids by age 50.
Uterine fibroids are stiff tumors that contain a significant amount of collagen and can become quite large. While they are non-cancerous, they can contribute to severe morbidity (pain, bleeding, anemia) and infertility.
Fibroids are the leading cause of hysterectomies in the U.S. Yet, many women want to keep their uterus. Existing minimally invasive surgical procedures that preserve the uterus are expen sive and are performed only at major medical centers. Reinterventions are commonly required (up to 50%) to treat regrown or new fibroids. Cur rently available drug therapies have significant systemic side effects.
The personal and medical management of these benign tumors costs tens of billions annually in the U.S. — more than twice the cost of breast cancer.
Summary of the Project
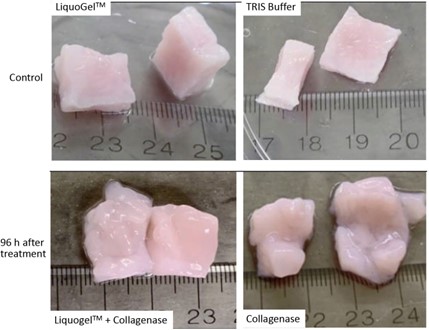
This project involved two aligned laboratory dis coveries. First, Duke’s scientists were able to significantly soften human fibroid tissue by injecting a highly purified enzyme that can degrade the collagen in fibroids ( Clostridium Histolyticum col lagenase (CHC)). Second, North Carolina Central University scientists pioneered the development of a breakthrough injectable hydrogel-copolymer (LiquoGel™ ) that allows delivery of a high drug payload; this can be single drugs or a cocktail drug mixture, and it can include and deliver wa-ter-soluble and non-water-soluble drugs. These two discoveries permitted novel testing of inject-able drug delivery to evaluate treatment for uterine fibroids by local injections.
To conduct this research, chemists at North Carolina Central University developed standard operating procedures for the preparation of larg er batches (20 g) of LiquoGel™ with purity in ex cess of 98% as confirmed by nuclear magnetic resonance spectroscopy. The LiquoGel™ carrier is liquid at room temperature and transitions to a gel at normal body temperatures. This protects locally delivered therapeutic drugs. Such drugs and other substances can be “entrapped” in the “pores” of the hydrogel-copolymer. The team was also able to develop batches of LiquoGel™ with more firmly (covalently) bound substances (LiquoGel™–R) that can also be loaded with other drugs. More firmly bound substances could be used as markers or “beacons” for treatment or diagnostic applications.
Working with Duke clinicians and scientists, human uterine fibroid tissue was then collected after routine surgery. In the laboratory, the team evaluated degradation/softening of these fibroid tissues at various time points after injection with CHC with and without LiquoGel™. To characterize and quantify the effects of local CHC injections on collagen, the microscopic structure of these tissues was examined and the stiffness modulus and viscoelasticity of the uterine fibroid tissues were measured. Injected fibroid tissues were ul-timately liquefied or exhibited gel-like behavior.
Summary of Results
The results of this intervention show promise for development of injectable and localized en-zymatic therapies for uterine fibroids and other dense tumors. In this pilot study, CHC injections reduced the tissue modulus by over two orders of magnitude and co-injection of LiquoGel™ en- hanced this effect.
This unprecedented approach to deliver drugs locally via LiquoGel™ can avoid systemic side ef- fects and treat uterine fibroid tumors. Leveraging this targeted technology could transform uterine fibroid therapy by offering a minimally invasive and non-surgical injectable alternative treatment that allows patients to keep their uterus thus re-ducing morbidity and costs associated with this disease.
Future Directions
Initial cytotoxicity studies have been done ex-vivo and in mice, but injection of the drug delivery poly- mer into a human cavity has to undergo in-vivo tri-als. Through a recently awarded NC Biotechnology TRG grant, the team is currently fine-tuning the parameters needed to scale up production of the polymer and to further test the added benefit of drug delivery in combination with LiquoGel™ .
Impact of the Project
Clinical and Medical
This project resulted in development, testing, and refinement of a drug delivery system that can be used in uterus-preserving, minimally invasive therapy for the treatment of uterine fibroids (or other dense tumors).
The delivery system can entrap or covalently bind a high payload and a wide range of drug class- es. It is liquid at room temperature and transitions to a gel at body temperature thereby reducing washout and localizing the drug to the place of injection. Collagen degrading enzymes or other drugs can be targeted to work locally, thereby re- ducing or eliminating drug systemic side effects.
Community
Fibroid treatment options are very limited, espe- cially for women with fibroids who want to still have children and for women at high risk for sur- gical complications. Many woman experience high morbidity (i.e. pain, bleeding, anemia, infertility) leading to reduced quality of life and decreased productivity. Society often allows women to suffer in silence and accept gynecologic pain as “normal”. Ultimately the uterus preserving, local injection of fibroids could be performed under ultrasound guidance in a doctor’s office. Women would not have to take the risk of surgical removal of their uterus and/or fibroids, avoid the hospital stay and long recovery, and preserve their uterus.
The availability of a new treatment option that is minimally invasive can decrease barriers for women to seek treatment at an earlier stage in their disease.
Economic
Uterine fibroids result in high healthcare costs and loss of productivity in affected women. The management of uterine fibroids is estimated at $34 billion annually in the U.S. Minimally invasive, local treatment of fibroids can offer a more ac- ceptable, less costly, and faster treatment option, thus decreasing women’s suffering and increasing their ability to live a productive life.
The developed and tested drug delivery platform can be licensed for use in fibroids and other diseas- es that would benefit from local drug delivery.
Workforce Development & Diversity
Darlene Taylor was a NIH K12 BIRCWH Scholar (06/01/2008–05/31/2013). These five years of BIRCWH support facilitated engagement in a suite of biological topics (i.e., five Clinical Research Training Program courses, Molecular Biology Workshop 2009, uterine fibroids, breast cancer tumors, etc.)
Together, her unique biological/polymer physi- cal training positioned Dr. Taylor to attract external funding ($471k), deliver eight (8) presentations, and publish her research in peer-reviewed journals. She was also able to: 1) Launch a quality polymer biomaterials initiative at NC Central that focuses on addressing women’s health through advanced polymer materials; 2) Obtain promotion and ten- ure to associate professor and receive the adjunct appointment at Duke and in BRITE (a research-in- tensive program at NC Central); and 3) pioneer the development of a novel drug delivery platform, namely LiquoGel™ . Now as a full professor in the Department of Chemistry and Biochemistry at NCCU, Dr. Taylor has built a network of research collaborators to pursue innovative delivery op-tions of therapeutic agents to tumors affecting the health of women.
Health Equity & Disparities
Black women have a greater disease burden and a hysterectomy rate for fibroids three-fold higher than White women. Black women develop uter- ine fibroids at an earlier age resulting in a great- er need to utilize uterus-preserving treatment modalities. A new minimally invasive treatment of fibroids would especially benefit these women who want to preserve their uterus. Local delivery under ultrasound guidance using IVF needles is feasible. Our results show promise for develop- ment of injectable and localized enzymatic thera- pies for uterine fibroids and other dense tumors.
Disease Estimated Annual Cost
- Diabetes: $193 billion
- Uterine Fibroids: $34 billion
- Breast Cancer: $16 billion
- Colon Cancer: $14 billion
- Ovarian Cancer: $5 billion
Source: Cardozo ER, Clark AD, Banks NK, Henne MB, Stegmann BJ, Segars JH. The estimated annual cost of uterine leiomyomata in the United States. Am J Obstet Gynecol. 2012 Mar;206(3):211.e1-9. doi: 10.1016/j. ajog.2011.12.002. Epub 2011 Dec 11. PMID: 22244472; PMCID: PMC3292655.
About the Research Team

Darlene Taylor, PhD
Principal Investigator
Department of Chemistry
North Carolina Central University

Friederike L. Jayes, PhD
Principal Investigator
Department of Obstetrics and Gynecology
Duke University School of Medicine
Robert Vachieri, Technician
Department of Chemistry,
North Carolina Central University
Tracy Truong, Biostatistician
Department of Biostatistics & Bioinformatics
Duke University School of Medicine
Emily Miller, Project Manager Duke CTSI,
Duke University School of Medicine
Project Milestones
| Date | Milestone | Description |
|---|---|---|
| JAN 2019 | Project Start and Funding | Received $56,131 funding from CTSI to conduct pilot studies. |
| DEC 2019 | Project End Date | Pilot project ends. Research continues. |
| FEB 2021 | Publication | First paper from pilot study published in Acta Biomaterialia |
| FEB 2021 | Invited Speaker | Results from pilot study shared at Fibroid Summit |
| FEB 2021 | Follow-on Funding |
Translational Research Grant from the NC Biotechnology Center: Scale up production of LiquoGel™ to facilitate IND-enabling experiments and the time course of drug activity after ex vivo injection into human tissue samples. Additionally, evaluate the stability and degradation products of optimized composition of LiquoGel™. |
| SEP 2022 | Invention Disclosure Form Submitted |
Additional components of the treatment system and formalize collaborations to further develop this novel treatment mechanism |
Translational Science Benefits Summary
Investigative Procedures
Refinement of an ex-vivo model for testing enzymatic degradation of extracellular matrix in human fibroids using injections, incubation, histology, and rheology. (Demonstrated)
Therapeutic Procedure
Development, testing, and refinement of a drug delivery system that can be used in uterus-preserv-ing, minimally invasive therapy for the treatment of uterine fibroids (or other dense tumors). Collagen degrading enzymes or other drugs can be targeted to work locally, thereby reducing or eliminating drug systemic side effects. (Potential)
Biomedical Technology
Development, testing, and refinement of a drug delivery system that can be used in local drug deliv-ery. The delivery system entraps or covalently binds a wide range of drug classes. It is liquid at room tem-perature and transitions to a gel at body tempera-ture. (Potential)
Products and Devices
Drug delivery system that can be used in local drug delivery. The delivery system can entrap or covalently bind a high payload and a wide range of drug classes.
It is liquid at room temperature and transitions to a gel at body temperature thereby reducing wash-out and localizing the drug to the place of injection. (Demonstrated)
License Agreements
The developed and tested drug delivery platform can be licensed for use in fibroids and other diseases that would benefit from local drug delivery. (Poten-tial)
Societal & Financial Cost of Illness
Uterine fibroids result in high healthcare costs and loss of productivity in affected women. The manage-ment of uterine fibroids is estimated at $34 billion annually in the U.S. Minimally invasive, local treat-ment of fibroids can offer a more acceptable, less costly, and faster treatment option, thus decreasing women’s suffering and increasing their ability to live a productive life. (Potential)
Health Care Delivery
The uterus-preserving, local injection of fibroids could be performed under ultrasound guidance in a doctor’s office. Women could preserve their uterus and avoid the risk of surgical removal of their uterus and/or fibroids, as well as a hospital stay and long recovery.
Since Black women usually develop fibroids earlier during their reproductive life, this patient population would especially benefit from such treatment option. (Potential)
Life Expectancy and Quality of Life
The availability of a new treatment option that is minimally invasive can decrease barriers for women to seek treatment at an earlier stage in their disease. (Potential)
CTSA Resources Used
| Core | Type of Service |
|---|---|
| CTSI Accelerator/Pilots | Funding and Project Manager Support |
| Biostatistics, Epidemiology, and Research Design (BERD) | Statistical Support |
| Regulatory Affairs | Consultation on long-term FDA approval planning for LiquoGel™ |
Other Institutional Resources Used
| Group | Type of Service |
|---|---|
| Duke BioRepository & Precision Pathology Center (BRPC) | Collected fibroid specimen from surgical pathology |
| Duke IRB | Reviewed and approved necessary changes/amendments |
| NIH Duke/NCCU BIRCWH K12 Career Development Award | Dr. Darlene Taylor was a 2008-2013 Scholar. During this time period she was promoted from Assistant to Associate Professor and received an Adjunct Professor appointment in the Department of OB/GYN at Duke. |
For More Information
Reference:
Corder RD, Gadi SV, Vachieri RB, Jayes FL, Cullen JM, Khan SA, Taylor DK. Using rheology to quantify the effects of localized collagenase treatments on uterine fibroid digestion, Acta Biomater. 2021;134:443-452. doi: 10.1016/j.actbio.2021.08.003
© Duke Clinical & Translational Science Institute June 2023